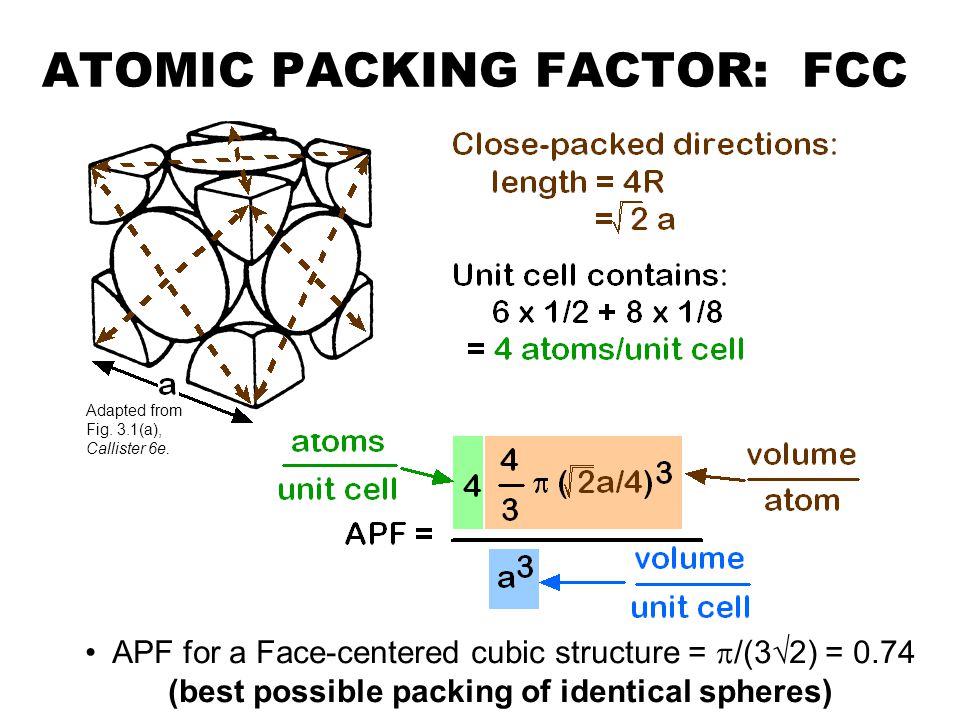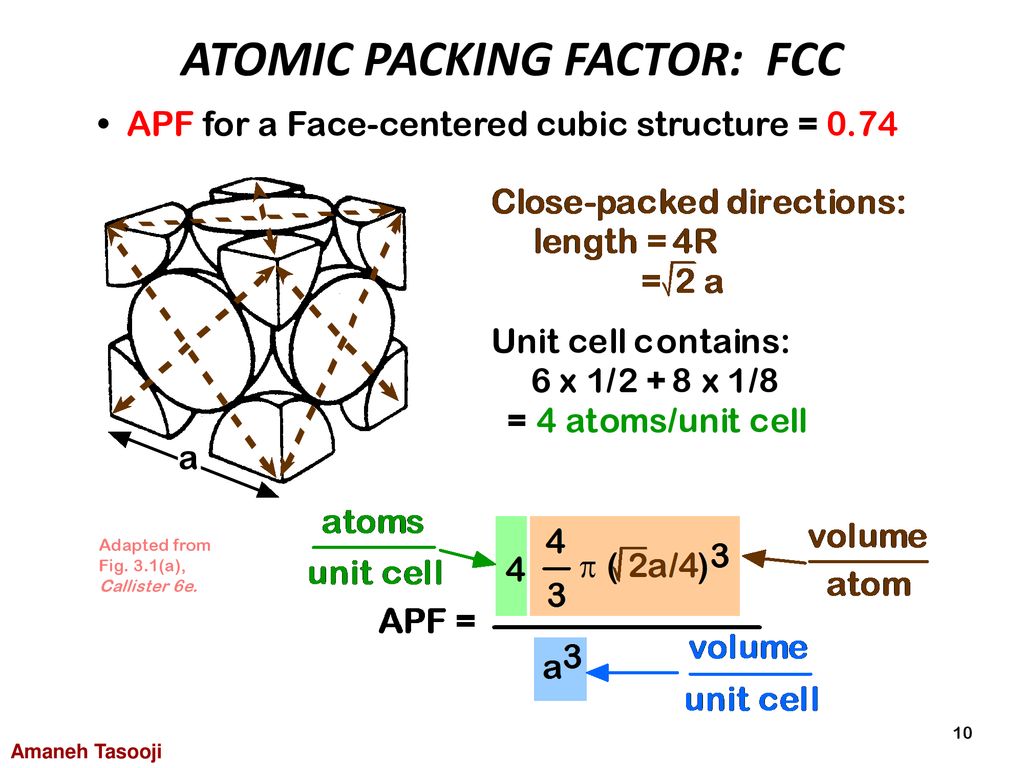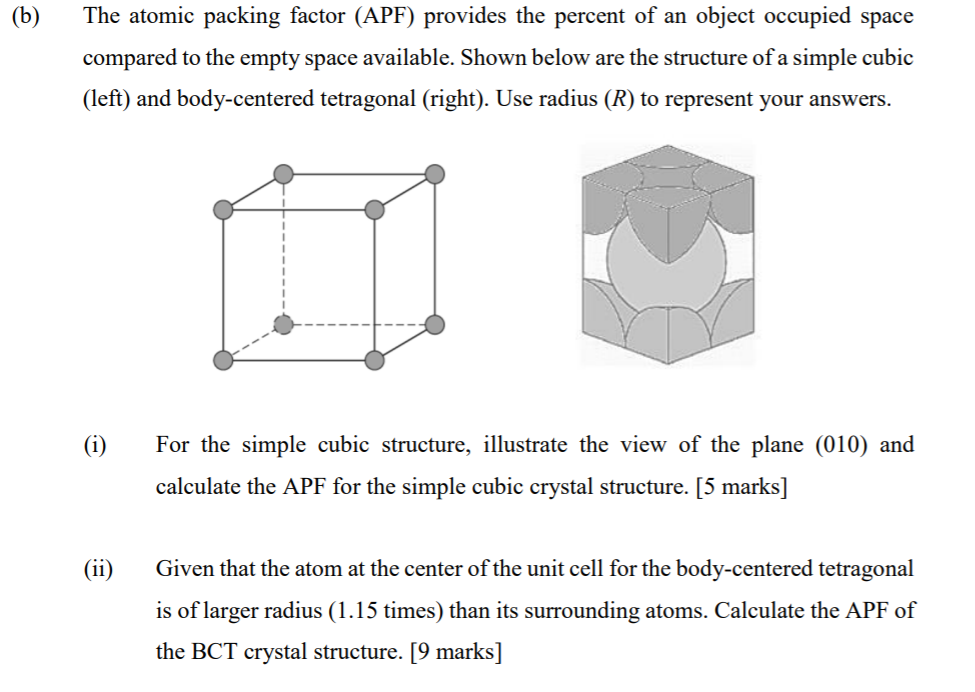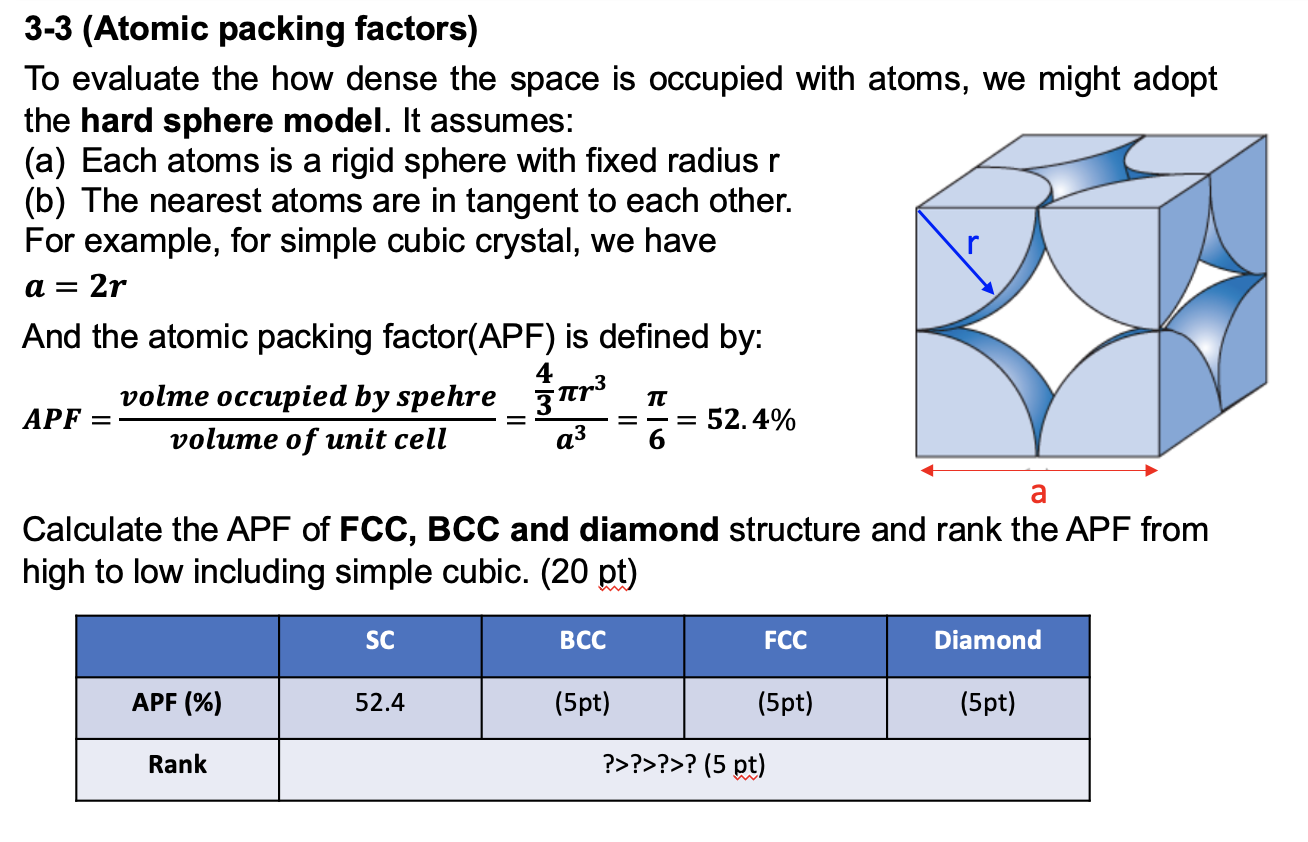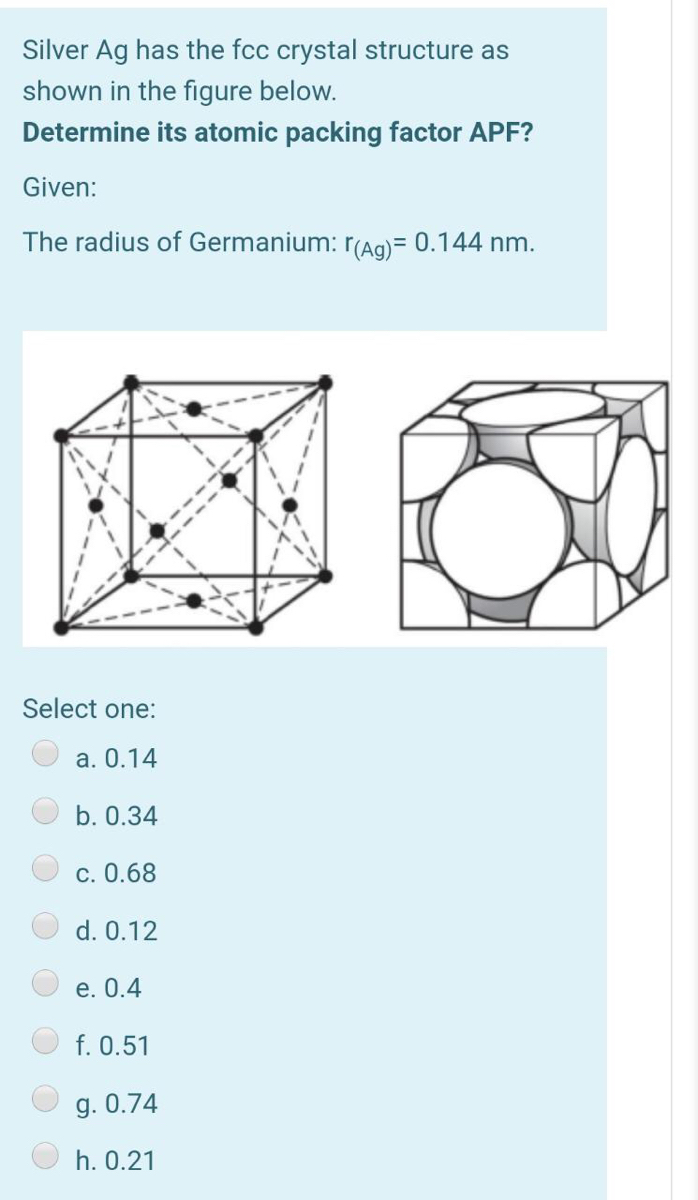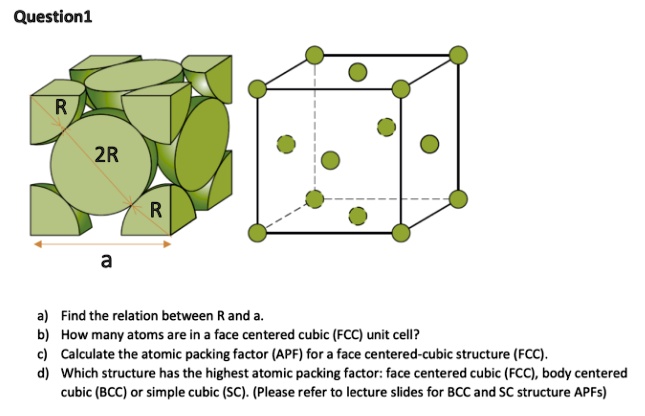
SOLVED: Question 1: Find the relation between R and how many atoms are in a face-centered cubic (FCC) unit cell. Calculate the atomic packing factor (APF) for the face-centered cubic structure (FCC).
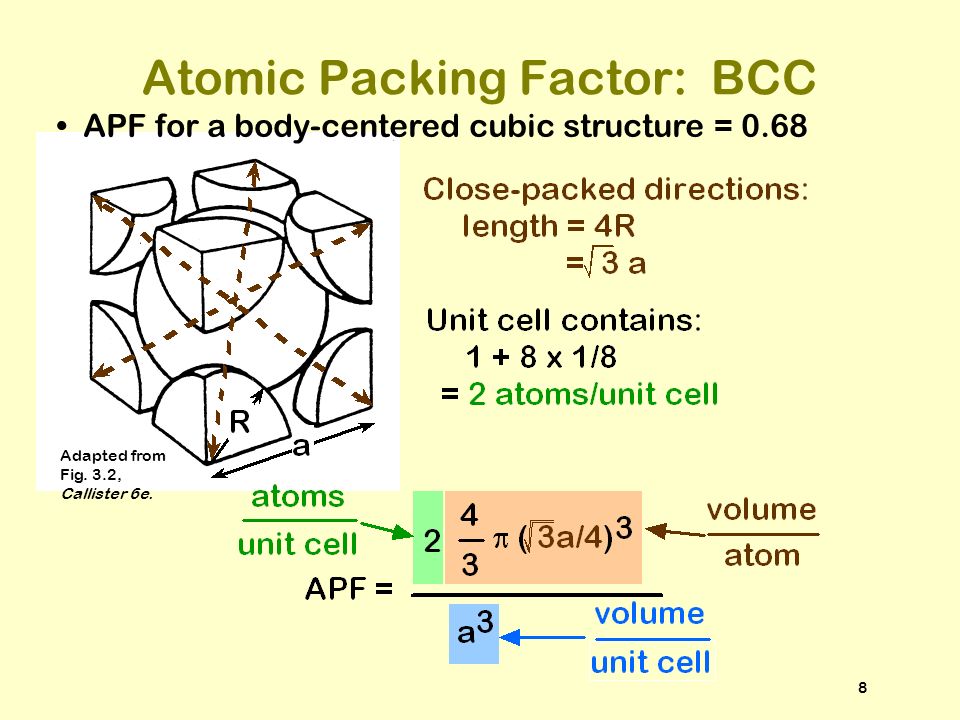
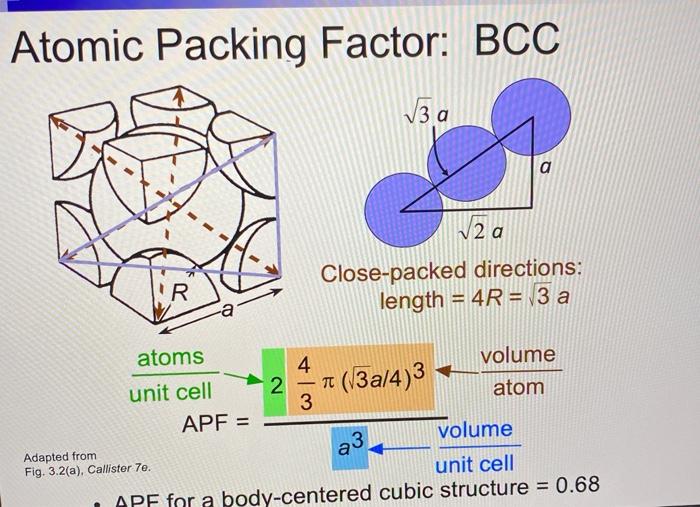
ISSUES TO ADDRESS... How do atoms assemble into solid structures? (for now, focus on metals) How does the density of a material depend on its structure? - ppt download

Finding Atomic Packing Factor or Packing Efficiency for Simple Cubic, BCC & FCC | Tamil - தமிழில் - YouTube
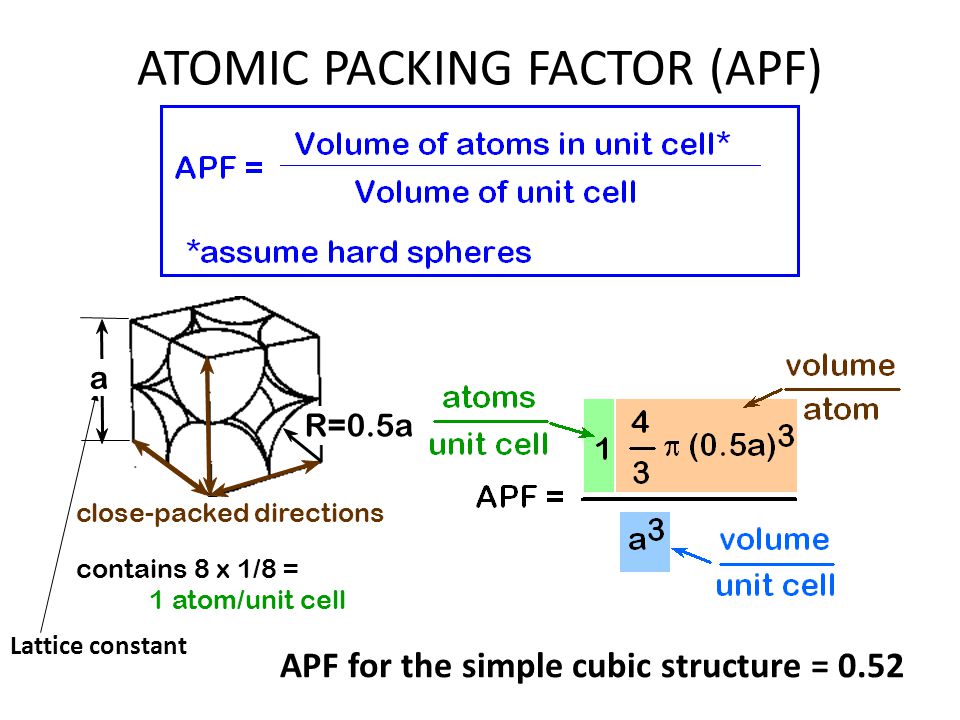
Structure of Solids Objectives By the end of this section you should be able to: Calculate atomic packing factors (HW) Compare bcc, fcc and hcp crystal. - ppt download