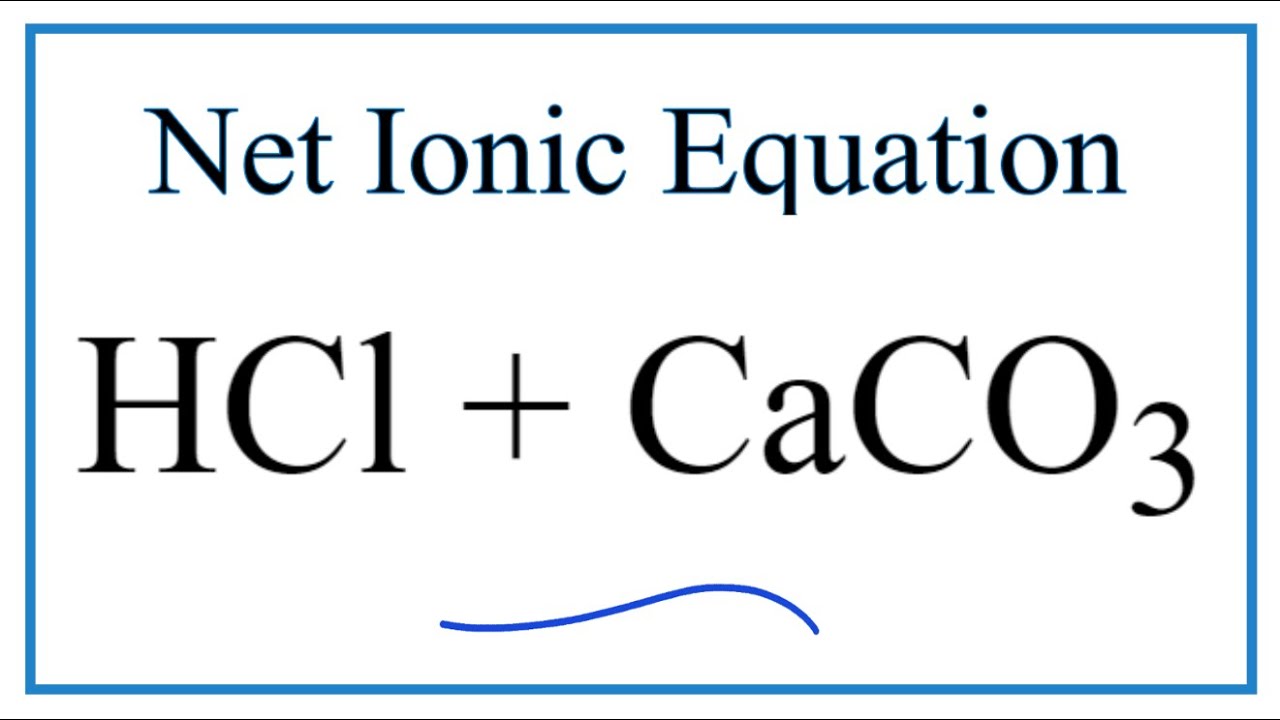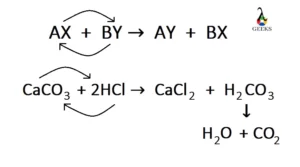How to Balance CaCO3 + HCl = CaCl2 + CO2 + H2O | How to Balance CaCO3 + HCl = CaCl2 + CO2 + H2O Hey there! Are you struggling with balancing

Balance the following equations: i) CaCO3 + HCl → CaCl2 + CO2 ↑ + H2O ii) Na + H2O → NaOH + H2 ↑ iii) (NH4)2 SO4 + Ca(OH)2 → CaSO4 +

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa
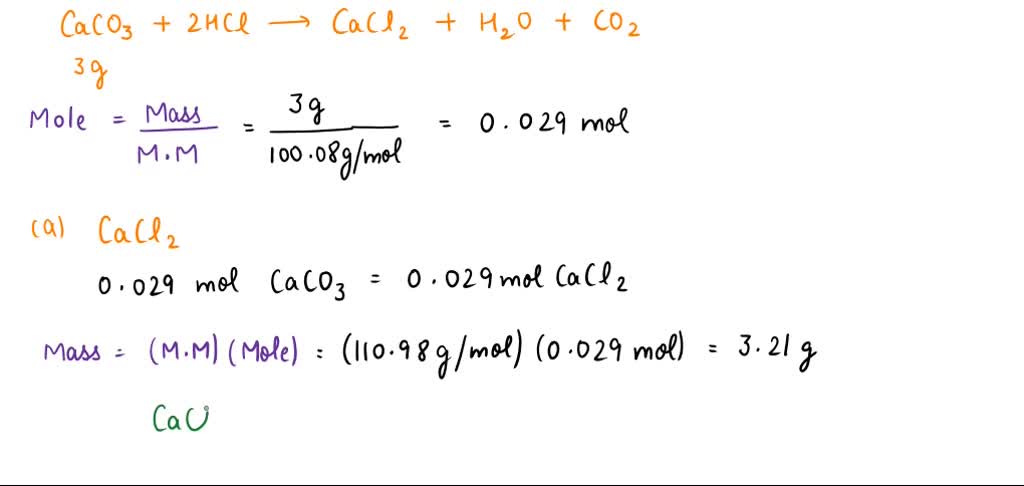
SOLVED: IF 3.00g of limestone reacted according to the equation CaCO3 + 2 HCl = CaCl 2 + H2O + CO2. What masses of the following would be produced? Calcium Chloride Carbon Dioxide Water

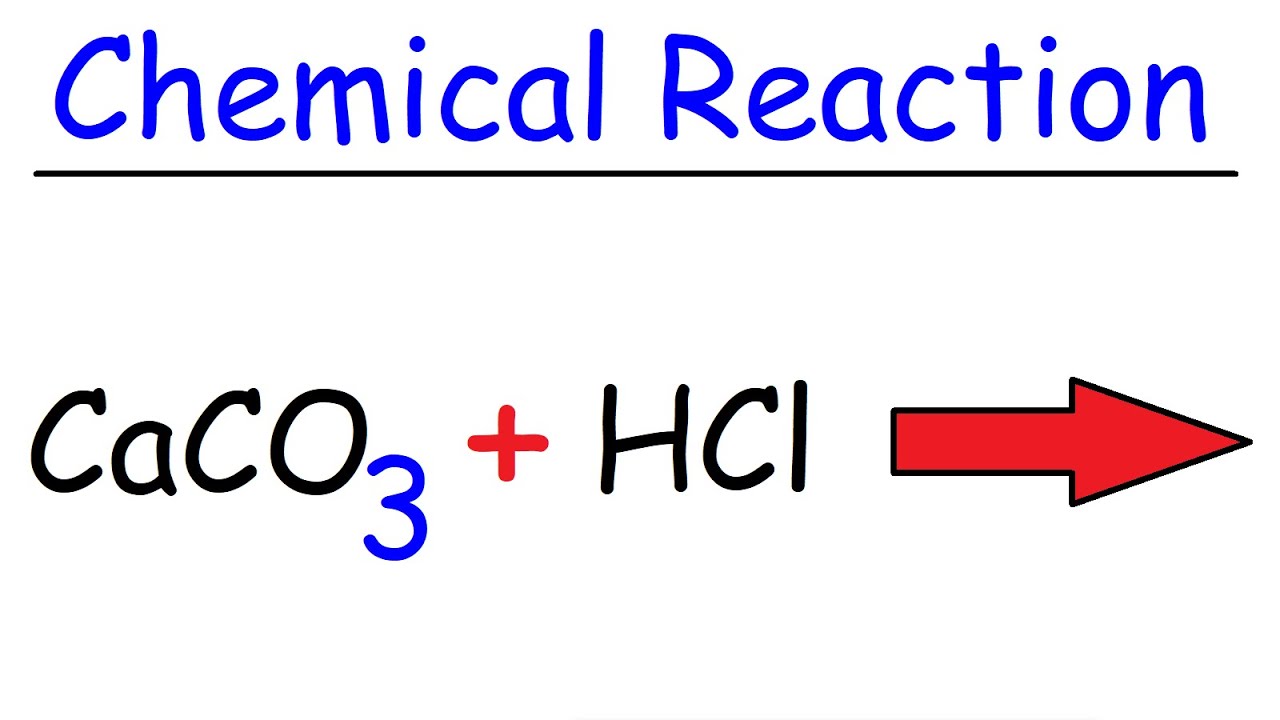
Write a balanced chemical equation for the reaction of calcium carbonate and dil. hydrochloric acid.
What mass of calcium carbonate can 2000. mL of a 1.50M aqueous HCl solution dissolve? 2 HCl(aq) + CaCO3(s) à CaCl2(aq) + CO2(g) + H2O(l) - Quora

Which of the following reactions is balanced? A.CaCl2 + 2H2CO3 = CaCO3 + HCI B.2C2Cl2 + H2CO3 +CaCO3 + - Brainly.com
16. In a chemical reaction, caco3+2hcl= cacl2 +co2+h2o. 25ml hcl and 0.75M Calculate the amount of caco3

Amount of dissolved calcium carbonate in different chemical reagent systems | Download Scientific Diagram
Complete and balance the following chemical equations: i. CaCO3 + HCl - Sarthaks eConnect | Largest Online Education Community