Consider the following reactions, C (s) + O2 (g) → CO2 (g), Δ H = - 94 kcal 2CO (g) + O2 → 2 CO2 (g) , Δ H = - 135.2 kcalThen, the heat of formation of CO (g) is:
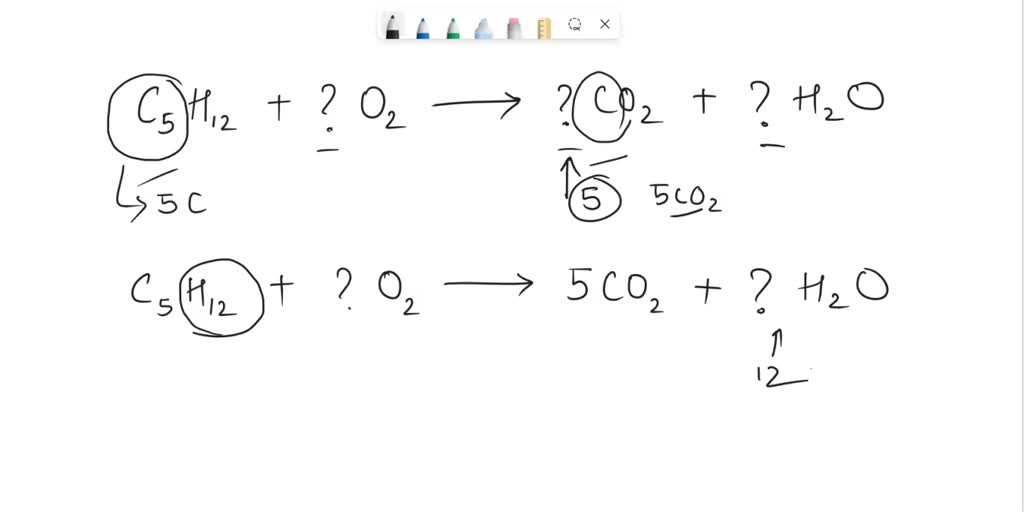
SOLVED: What is the coefficient for carbon dioxide in the balanced equation? C5H12 + ? O2 → ? CO2 + ? H2O A) 2 B) 4 C) 5 D) 6 E) 8
Given that, C + O2 → CO2; ΔH° = -x kJ, 2CO + O2 → 2CO2; ΔH° = -y kJ. The enthalpy of formation of CO will be - Sarthaks eConnect | Largest Online Education Community